Dissolution testing is how the FDA ensures generic drugs release active ingredients at the same rate as brand-name versions, without requiring human trials. This science-based method keeps generics safe, effective, and affordable.
Tag: bioequivalence
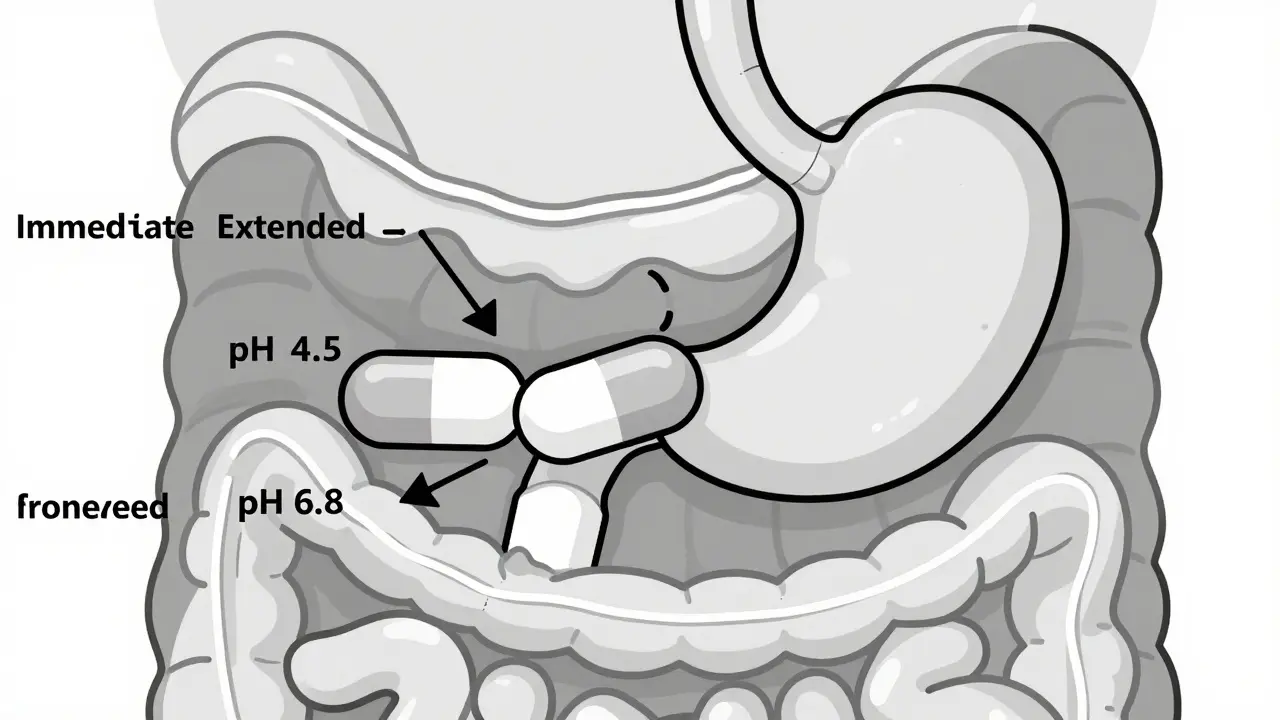
Modified-release formulations require specialized bioequivalence testing to ensure generics work as well as brand-name drugs. Learn how regulators assess release profiles, dissolution, and partial AUCs-and why some generics still fail patients.